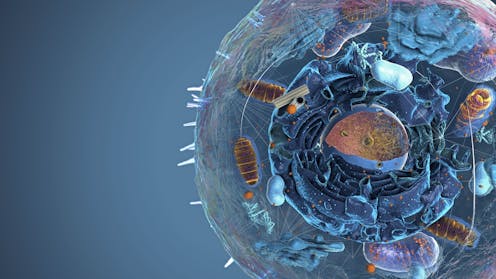
Think back to that basic biology class you took in high school. You probably learned about organelles, those little “organs” inside cells that form compartments with individual functions. For example, mitochondria produce energy, lysosomes recycle waste and the nucleus stores DNA. Although each organelle has a different function, they are similar in that every one is wrapped up in a membrane.
Membrane-bound organelles were the textbook standard of how scientists thought cells were organized until they realized in the mid-2000s that some organelles don’t need to be wrapped in a membrane. Since then, researchers have discovered many additional membraneless organelles that have significantly changed how biologists think about the chemistry and origins of life.
I was introduced to membraneless organelles, formally called biomolecular condensates, a couple years ago when students in my lab observed some unusual blobs in a cell nucleus. Unbeknownst to me, we had actually been studying biomolecular condensates for years. What I finally saw in those blobs opened my eyes to a whole new world of cell biology.
Like a lava lamp
To get a sense of what a biomolecular condensate looks like, imagine a lava lamp as the blobs of wax inside fuse together, break apart and fuse again. Condensates form in much the same way, though they are not made of wax. Instead, a cluster of proteins and genetic material, specifically RNA molecules, in a cell condenses into gel-like droplets.
Some proteins and RNAs do this because they preferentially interact with each other instead of their surrounding environment, very much like how wax blobs in a lava lamp mix with each other but not the surrounding liquid. These condensates create a new microenvironment that attracts additional proteins and RNA molecules, thus forming a unique biochemical compartment within cells.
As of 2022, researchers have found about 30 kinds of these membraneless biomolecular condensates. In comparison, there are around a dozen known traditional membrane-bound organelles.
Although easy to identify once you know what you are looking for, it’s difficult to figure out what biomolecular condensates exactly do. Some have well-defined roles, such as forming reproductive cells, stress granules and protein-making ribosomes. However, many others don’t have clear functions.
Nonmembrane-bound organelles could have more numerous and diverse functions than their membrane-bound counterparts. Learning about these unknown functions is affecting scientists’ fundamental understanding of how cells work.
Protein structure and function
Biomolecular condensates are breaking some long-held beliefs about protein chemistry.
Ever since scientists first got a good look at the structure of the protein myoglobin in the 1950s, it was clear that its structure is important for its ability to shuttle oxygen in muscles. Since then, the mantra for biochemists has been that protein structure equals protein function. Basically, proteins have certain shapes that allow them to perform their jobs.
The proteins that form biomolecular condensates at least partially break this rule since they contain regions that are disordered, meaning they do not have defined shapes. When researchers discovered these so-called intrinsically disordered proteins, or IDPs, in the early 1980s, they were initially confounded by how these proteins could lack a strong structure but still perform specific functions.
Later, they found that IDPs tend to form condensates. As is so often the case in science, this finding solved one mystery about the roles these unstructured rogue proteins play in the cell only to open another deeper question about what biomolecular condensates really are.
Bacterial cells
Researchers have also detected biomolecular condensates in prokaryotic, or bacterial, cells, which traditionally were defined as not containing organelles. This finding could have profound effects on how scientists understand the biology of prokaryotic cells.
Only about 6% of bacterial proteins have disordered regions lacking structure, compared with 30% to 40% of eukaryotic, or nonbacterial, proteins. But scientists have found several biomolecular condensates in prokaryotic cells that are involved a variety of cellular functions, including making and breaking down RNAs.
The presence of biomolecular condensates in bacterial cells means that these microbes aren’t simple bags of proteins and nucleic acids but are actually more complex than previously recognized.
Origins of life
Biomolecular condensates are also changing how scientists think about the origins of life on Earth.
There is ample evidence that nucleotides, the building blocks of RNA and DNA, can very plausibly be made from common chemicals, like hydrogen cyanide and water, in the presence of common energy sources, like ultraviolet light or high temperatures, on universally common minerals, like silica and iron clay.
There is also evidence that individual nucleotides can spontaneously assemble into chains to make RNA. This is a crucial step in the RNA world hypothesis, which postulates that the first “lifeforms” on Earth were strands of RNAs.
A major question is how these RNA molecules might have evolved mechanisms to replicate themselves and organize into a protocell. Because all known life is enclosed in membranes, researchers studying the origin of life have mostly assumed that membranes would also need to encapsulate these RNAs. This would require synthesizing the lipids, or fats, that make up membranes. However, the materials needed to make lipids likely weren’t present on early Earth.
With the discovery that RNAs can spontaneously form biomolecular condensates, lipids wouldn’t be needed to form protocells. If RNAs were able to aggregate into biomolecular condensates on their own, it becomes even more plausible that living molecules arose from nonliving chemicals on Earth.
New treatments
For me and other scientists studying biomolecular condensates, it is exciting to dream of how these rule-breaking entities will change our perspective on how biology works. Condensates are already changing how we think about human diseases like Alzheimer’s, Huntington’s and Lou Gehrig’s.
To this end, researchers are developing several new approaches to manipulate condensates for medical purposes like new drugs that can promote or dissolve condensates. Whether this new approach to treating disease will bear fruit remains to be determined.
In the long term, I wouldn’t be surprised if each biomolecular condensate is eventually assigned a particular function. If this happens, you can bet that high school biology students will have even more to learn – or complain – about in their introductory biology classes.
Allan Albig receives funding from the National Institute of Health.
This article was originally published on The Conversation. Read the original article.