Pfizer (NYSE:PFE) and its partner, BioNTech (NASDAQ:BNTX) announced that they have completed the submission of a regulatory application to the European Medicines Agency (EMA), which seeks authorization for their Omicron-adapted bivalent COVID-19 vaccine candidate in individuals aged 12 years and older.
The bivalent vaccine is a combination of Pfizer/BioNTech's currently marketed COVID-19 vaccine, Comirnaty and a vaccine candidate targeting the spike protein of the Omicron BA.1 subvariant. The regulatory filing by Pfizer and BioNTech follows the guidelines laid down by both the EMA and the International Coalition of Medicines Regulatory Authorities to develop Omicron-specific vaccines for tackling the problem of new and evolving COVID-19 variants.
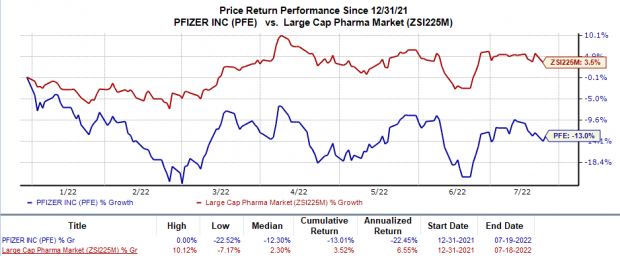
Shares of Pfizer have lost 13% in the year so far against the industry's 3.5% rise.
The filing with the EMA is based on data from a phase II/III study announced last month that evaluated the safety, tolerability, and immunogenicity of two Omicron-adapted COVID-19 vaccine candidates, one monovalent and the other bivalent.
Data from the phase II/III study showed that participants given the booster dose of both the vaccine candidates generated a substantially higher immune response against the Omicron subvariant BA.1 when compared to Comirnaty.
Following one month of dose administration, participants who received the 30 µg dose of the bivalent vaccine candidate achieved a 9.1-fold increase in neutralizing geometric mean titers when compared to those who received Comirnaty.
Pfizer and BioNTech have already shared the above data with the FDA to support the authorization of the bivalent vaccine candidate. The regulatory body has also requested the companies to share additional data on the candidate.
Comirnaty is currently authorized in the European Union for use in individuals aged 5 years and older. A third/booster dose of the vaccine has been authorized for use in individuals aged 12 years and older.
The COVID-19 vaccines developed by Pfizer and BioNTech are based on mRNA technology. Another vaccine based on mRNA technology, which poses stiff competition to the Pfizer/BioNTech vaccine, has been developed by Moderna MRNA.
Moderna is also working on developing a new Omicron-based COVID-19 vaccine. Last month, Moderna announced data from an ongoing phase II/III study, which evaluated its bivalent COVID-19 vaccine booster candidate, mRNA-1273.214, targeting the Omicron variant.
The clinical data provided by Moderna showed that following one month of administration of the 50 µg dose of mRNA-1273.214, a 5.4-fold increase in neutralizing antibodies against the Omicron subvariants BA.4 and BA.5 was seen in all study participants, regardless of prior infection. In a subset of seronegative participants, the candidate generated a 6.3-fold increase in neutralizing titers against both the Omicron subvariants. Based on the above results, Moderna intends to complete the regulatory filing for mRNA-1273.214 in the coming weeks.
Zacks Rank & Stock to Consider
Pfizer currently carries a Zacks Rank #3 (Hold). A better-ranked stock in the same sector is Merck MRK, which carries a Zacks Rank #1 (Strong Buy) at present.
Estimates for Merck's 2022 bottom line have increased from $7.28 to $7.31 in the past 30 days. Shares of MRK have risen 20.5% in the year-to-date period.
Earnings of Merck beat estimates in three of the last four quarters and missed the mark on one occasion, the average surprise being 13.4%. In the last reported quarter, MRK delivered an earnings surprise of 18.2%.
To read this article on Zacks.com click here.
Zacks Investment Research









