Scientists have unveiled a detailed "atlas" of the placenta and uterus, showing how these unique tissues grow and evolve throughout pregnancy to accommodate a developing fetus.
In charting this new map, the scientists revealed a subtype of cell that had never been described before and appears to be unique to pregnancy.
These cells are not present in the uterus outside pregnancy, and they suddenly rise in number at the start of gestation as the uterine lining morphs to cradle and support the embryo, explained study first author Cheng Wang, a specialist in regeneration medicine specialist at the University of California, San Francisco (UCSF).
"That was an exciting moment during the study," study senior author Jingjing Li, an associate professor of neurology at UCSF who studies human genomics, said of the cells' discovery. "We asked around — no one knows what they are."
These newly described cells seem to be involved in linking the placenta to the maternal blood supply, and they carry receptors that respond to cannabinoids. Cannabinoids include body-made chemicals, as well as the cannabis compounds THC and CBD. Therefore, the researchers suspect these cells may help to explain why cannabis use in pregnancy is tied to health consequences such as decreased blood flow to the placenta; poor oxygen delivery to the fetus; and a heightened risk of preterm birth, low birth weight and NICU admission.
It's unlikely that these cells' sensitivity to cannabinoids fully explains the risks posed by cannabis use in pregnancy, Li told Live Science; other potential culprits have been described in the medical literature. Nonetheless, these newfound cells are a factor that warrant further study, he said.
A crucial "speed bump"
Prior to the new study, published April 8 in the journal Nature, other research groups had mapped the placenta and uterus using similar techniques. However, those previous studies covered only select chapters of pregnancy.
"The biggest difference is we are looking at the whole time course" from early pregnancy to birth, Li said. The new atlas incorporates data from tissues that were collected between weeks 5 and 39 of pregnancy and then stored in tissue banks at UCSF and Stanford University.
Li's lab analyzes tissues in great detail, at the resolution of single cells, with placental development being one of the team's major research focuses. Their new atlas incorporates snapshots of which genes were active and which proteins were present in the analyzed cells at a given stage of pregnancy. It also looks at "chromatin accessibility," which reflects how DNA molecules are packaged within the cell and which genes can be activated at a given moment.
In total, the team analyzed about 1.2 million placental and uterine cells, including 200,000 isolated cells and 1 million cells embedded in their original locations within the tissue.
Looking at isolated cells can be helpful for pinpointing their identities, but the approach lacks the context of "where these cells are located within the tissue, which cells they are near, and how they interact with one another," Roser Vento-Tormo, a group leader in cellular genomics at the Wellcome Sanger Institute, and her postdoc Ana Paredes told Live Science via email. In the uterus and placenta, cells are arranged in distinct regions with different functions.
By combining these two techniques, "this study provides a clear example of how cell location and state can yield important biological insights into how a healthy pregnancy is orchestrated at the cellular and molecular level," said Vento-Tormo and Paredes, who were not involved in the research.
The work revealed interesting links between a given cell's gene activity and its behavior.
For instance, early in pregnancy, certain fetal cells invade the uterus and its major arteries, helping to establish blood flow to the placenta. Using machine learning, the researchers predicted how deeply a given cell would invade the uterus based on its gene activity.
When this invasion goes awry — for example, if cells do not penetrate deeply enough or they penetrate too deeply — it can contribute to complications like preeclampsia or placenta accreta, Vento-Tormo and Paredes said. "For a healthy pregnancy, this invasion needs to be very precisely controlled," they noted.
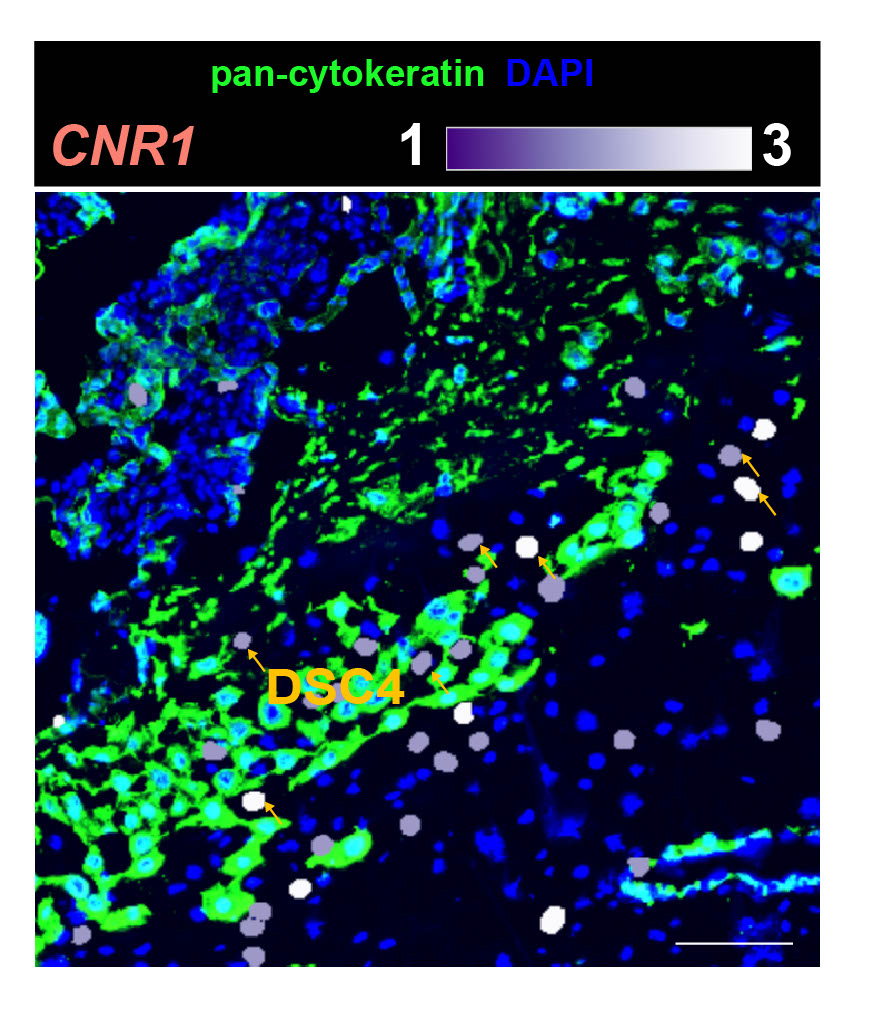
It turns out that the new cell type identified by the researchers helps to regulate the invasion. By sending out specific signals, the cell type acts as a "speed bump" to prevent the process from proceeding too quickly, Li said.
"It's at the frontline of the maternal-fetal interface," Weng told Live Science. Various proteins carried by these cells support this idea that they're regulating the behavior of other cells at this crucial interface, he said.
Future research could address whether changes in this signaling contributes to pregnancy disorders, as well as whether there may be treatment opportunities in targeting the pathway, Vento-Tormo and Paredes suggested.
With their completed map in hand, the researchers married their findings with data from huge genetics studies of preeclampsia, preterm birth and pregnancy loss. Those published studies had uncovered links between specific gene variants and the risk of these complications. The team could then pinpoint the specific cells in the placenta and uterus that actively use those genes and are therefore most vulnerable to the conditions.
"The question is, 'In which cell type will those high-risk variants take effect?'" Li said. "This will help us to know which cells are underlying those complications" and potentially develop treatments that target those cells in the future.
While the study brings together a trove of data, Li emphasized that there's more work to be done. The study focused on healthy pregnancies, so there's still a question of how pregnancies impacted by various conditions differ from this baseline. The team is now working with clinical partners to start making those comparisons. Overall, they aim to increase the total number of cells analyzed to make sure they're capturing the full diversity of cells in the pregnant uterus.
Vento-Tormo and Paredes suggested the new atlas could be combined with existing datasets, including one of their own, to help refine scientists' understanding of specific placenta cell types and their regulators. Some of the study's key findings could also be checked in laboratory models of the placenta.
"If we include more cells, more samples, a lot of new, exciting discoveries could be made," Li said. "So this is really a starting point."
Editor's note: This story was updated on April 9, 2026, to add comments from Ana Paredes and Roser Vento-Tormo.
This article is for informational purposes only and is not meant to offer medical advice.








